What is Alzheimer's disease?
Dementia, also called major neurocognitive disorder, encompasses a set of concurrent symptoms whose main symptom is a severe cognitive impairment that affects the functioning and activities of daily living of people who suffer from it.
The disease of Alzheimer's is the most common form of dementia (between 60-80% of cases). It is considered a neurocognitive disease of aging since its prevalence in Western populations increases from 1% among individuals aged 65 years to 25% among individuals over 90 years of age. Given the continuing aging of the population, Alzheimer's disease has been listed as the epidemic of the XXI century, being one of the main priorities at the public health level.
At the cognitive level, Alzheimer's disease is characterized by problems in memory, difficulties in recognizing or remembering the names of things (anomie), orientation problems, and deterioration in reasoning and decision-making skills. The symptoms at the beginning of the disease are subtle and progressively worsen to the point of interfering with daily tasks. Therefore, the cognitive deficits of Alzheimer's disease vary depending on the stage of the disease:
- Symptoms of mild Alzheimer's disease: This is the early and mild stage of Alzheimer's disease. The person is able to function independently. However, the person has episodes of mild memory loss or even the location of objects used on a day-to-day basis.
- Symptoms of severe Alzheimer's disease: This is the most serious stage. Communication becomes very difficult, and changes in physical abilities are experienced, including the ability to speak, sit and eventually swallow. The person cannot function on his own and loses track of recent experiences, memories, and events surrounding him.
- Symptoms of severe Alzheimer's diseaseThis is the most severe stage. Communication becomes very difficult, changes in physical abilities are experienced, including the ability to speak, sit and eventually swallow. The person is not able to function on his own and loses track of recent experiences, memories and events around him.

Genetic architecture of Alzheimer's disease
From a genetic point of view, Alzheimer's can be divided into two subgroups:
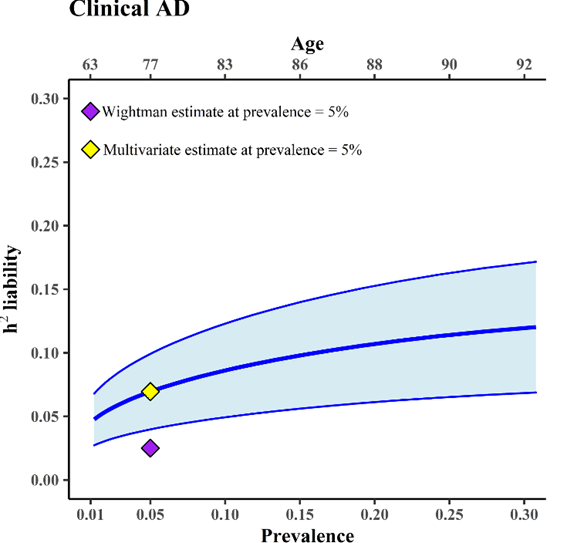
- Late onset or sporadic casesHeritability: this is the most common subgroup (90-95% of cases) influenced by multiple common variants whose individual effect on the disease is low. Heritability (proportion of variation of a trait in a population that can be attributed to genetic factors) is between 70-80% and its analysis is possible with genotyping (genomic technology used in the DNA TEST of ADNTRO).
- Early onset or familial casesEarly onset Alzheimer's disease is rare (5-10% of cases), occurring in people under 65 years of age, and the disease is most common in people under 65 years of age. heritabilityin this case, it is around 90%. It is usually explained by rare genetic variants that have a strong effect on the disease. Its analysis is carried out with the sequencing of the complete genome (service available at ADNTRO).
When we talk about genetic variants and effects, we find the following trend.
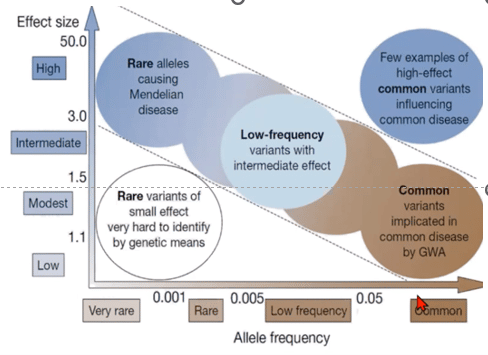
There are rare variants that can cause the early onset of Alzheimer's disease, for example: rare variants in the PSEN1, PSEN2 and APP genes. On the contrary, common variants (frequent in the population) alone tend to have less influence on the disease (the effect is smaller) making neccesary the existence of multiple common variants associated with the disease in order to present a high predisposition to develop the disease.
In ADNTRO we analyze genetic biomarkers associated with late-onset Alzheimer's, thus analyzing common genetic variants highly associated with Alzheimer’s Disease.
Your genetic predisposition to developing Alzheimer's
At ADNTRO, we analyze common genetic biomarkers associated with late-onset Alzheimer's disease, based on findings from large-scale genome-wide association studies (GWAS) conducted in large biobanks..
To calculate your predisposition or protection against Alzheimer's disease, we rely on a recent scientific study in which they identify more than two thousand common genetic variants associated with this disease.
Get to know your genetic predisposition to Alzheimer's disease in ADNTRO!
The genetics behind Alzheimer's disease
Genetic risk variants linked to Alzheimer's disease increase the risk of developing the disease in probabilistic terms, so high genetic risk for developing Alzheimer's disease does not mean that you will develop it. While it is true that being a homozygous carrier of the APOE-ɛ4 haplotype is associated with a risk of developing Alzheimer's disease between 8 and 10 times higher than non-carriers of this form of the gene (genetic variation in this gene explains approximately 10% of the genetic risk for Alzheimer's disease).
Genetic variants are specific points of DNA and therefore belong to or are very close to certain genes.
The most relevant genes for Alzheimer's disease are:
- CLU: helps regulate the removal of amyloid beta from the brain.
- CR1: a deficiency of the protein encoded by this gene can contribute to chronic inflammation of the brain
- PICALM: involved in the synapse. A correct synapse is essential for the proper functioning of neurons and the formation of memory.
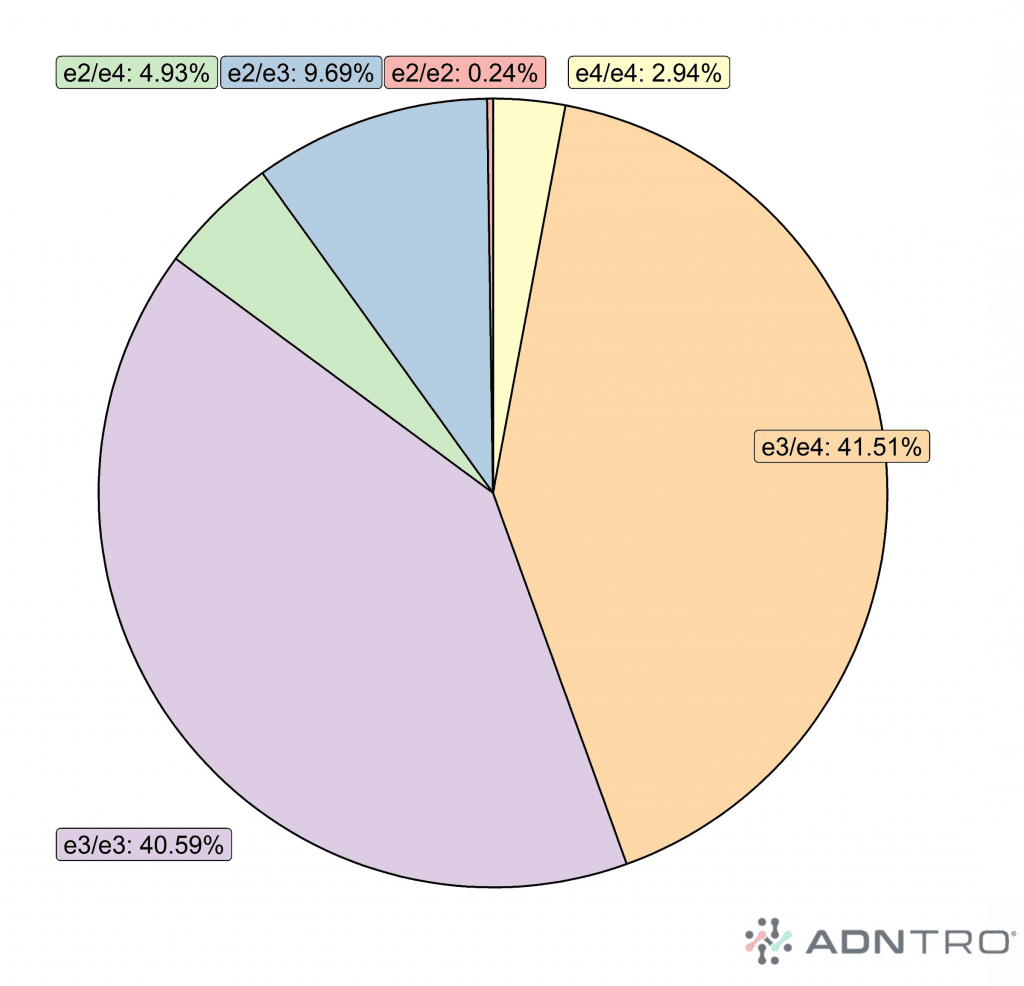
- APOE: is a highly studied and related gene to Alzheimer's disease. There are different "versions" (alleles) of this gene. Different combinations of these versions confer a greater or lesser risk of developing the disease. For this reason, in addition to obtaining your genetic predisposition based on more than 2,000 genetic variants associated with the risk of being diagnosed with Alzheimer's disease, we indicate your APOE haplotype. That is your "combination of possible versions of APOE". The APOE haplotype is based on the combination of the genotype present for two very specific genetic variants of the APOE gene. Discover your APOE haplotype with ADNTRO.
Your APOE haplotype
The APOE gene is a highly studied gene linked to Alzheimer's disease. Two common genetic variants within this gene determine the different ways that give a greater or lesser risk of developing the disease. For example, being a carrier of the APOE-ε4/APOE-ε4 form has been associated with an 8- to 10-fold increased risk of developing the disease. Inheriting the APOE ε4 setting does not mean that you will develop the disease, only that you have an increased risk for it.
The same applies the other way around. The fact of not having the APOE ε4 configuration does not exclude the possibility of developing Alzheimer's disease. For this reason, both the APOE haplotype and the "bar" with your result that we provide in ADNTRO are independent variables that indicate whether you have a higher or lower risk of developing the disease.
The genetic risk of Alzheimer's depends on many other genetic variants, with APOE being responsible for only 10% of the total risk.
The APOE haplotype explains approximately 10% of the genetic risk explained by common genetic variants of Alzheimer's disease, which represents a great effect for this region located on chromosome 19. It is estimated that the remaining common genetic risk variants associated with Alzheimer's disease are responsible for approximately another 10% of the genetic risk of it, leaving up to 80% of the genetic risk of Alzheimer's still undiscovered.
There are three configurations of the APOE gene (ε2, ε3, or ε4). Discover your APOE haplotype in ADNTRO!
Pathophysiology of Alzheimer's disease
The first phase of Alzheimer's disease is based on the death of cells in the hippocampus, the region of the brain where memories are stored. It also affects the parahippocampal cortex that is responsible for processes as important as memory and learning, thus losing the ability to store new memories.
Subsequently, the deterioration will expand to other brain structures such as the entorhinal cortex that performs extraordinarily important functions such as the consolidation of autobiographical memory, which allows us to know who we are.
Alzheimer's disease is clinically characterized by cognitive impairment (loss and death of neurons) that interferes with independence in everyday activities. And you may wonder, what causes all this damage to the brain? If we talk about Alzheimer's, at the molecular level it is worth highlighting three major characteristics responsible for the cognitive deterioration that characterizes the disease biologically:
- Formation of neurofibrillary tangles. These tangles appear from tau protein. This protein is responsible for stabilizing structures in our brain. The problem comes when the tau protein loses its stabilizing activity and forms precipitates (neurofibrillary tangles), which causes damage to the structure of our brain.
- Appearance of neuritic plaques. The person responsible for the formation of these plaques is a protein called beta-amyloid. When these precipitates are formed, a strong inflammatory response is triggered, thus generating damage to our brain. The protein encoded by the APOE gene is responsible for breaking these beta-amyloid plaques. It has been seen that the ε4 allele is less effective when performing this action, thus giving a greater risk of suffering from Alzheimer's disease to those who carry it.
- Loss of functional synapses. The synapse is the way our neurons communicate. To make it a little more visual and develop a simple molecular explanation, let's imagine a huge spider web created in the corner of a wall. If an ant falls into the spider's web, the structure remains intact, but in case of falling an entire anthill at once, the structure will be broken, eliminating the connections that existed in the first place between one end and the other of the wall. The same goes for our brain. All the connections formed between our neurons (synapses) can be altered and even destroyed with the formation of neurofibrillary tangles and neuritic plaques that are nothing more than precipitates (groups of ants) falling on our neural connections (spider web).
Recent studies suggest that the amyloid-beta protein is not an anomalous protein, but an endogenous molecule which is part of the brain's immune system. This positions the beta-amyloid protein as a key element of the brain's immune response when, for example, a brain trauma or bacterial infection occurs.
Due to the great similarity between the fat molecules that make up the membranes of bacteria and the membranes of brain cells, it is postulated that in cases of Alzheimer's disease, beta-amyloid might not distinguish between invading bacteria and brain cells, mistakenly attacking the latter.
In this case, if the new hypothesis is correct, Alzheimer's disease would be considered an autoimmune disease since the cause of the disease is due to a misdirected attack by the brain's immune system.
At ADNTRO we continue to investigate: towards a more accurate prediction of late-onset Alzheimer's.
Genomic research is advancing rapidly, and at ADNTRO we are actively contributing to that progress. Our team has published a study in the scientific journal Genes titled Development of a k-Nearest Neighbors Model for the Prediction of Late-Onset Alzheimer's Risk by Combining Polygenic Risk Scores and Phenotypic Variables, in which we developed a machine learning model capable of identifying people at risk of developing late-onset Alzheimer's even before the first symptoms appear.
The model combines genetic information, APOE configuration, and readily available clinical variables such as age, cholesterol, or diabetes history, achieving a sensitivity of 80% —compared to the 63% of previous comparable models—, which represents a significant advance in the early detection of this disease.
It's important to clarify that this research model, by incorporating clinical and personal data, has not been integrated into the ADNTRO platform, where we work exclusively with genetic information for privacy reasons. In your profile, we calculate your APOE configuration, one of the most determining markers in the risk of developing Alzheimer's, and also relevant for evaluating the response to treatments such as lecanemab, recently approved by the European Commission.
Publishing this type of research reflects our commitment to science: we not only offer a genetic test, but we also contribute to building the tools of the future in predictive medicine.
Sources and links of interest
https://www.fundacioace.com/es
https://www.nia.nih.gov/health/alzheimers-disease-genetics-fact-sheet
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9200312/