In recent years, thanks to the development of genomic techniques for massive sequencing and analysis with new bioinformatics tools, many biomarkers have been identified that are associated with how our body reacts to drugs. This is where we must define the concepts of pharmacokinetics and pharmacodynamics. Pharmacokinetics is the process of absorption, distribution, metabolism and excretion (ADME) of a drug and pharmacodynamics refers to the mechanism of action of a drug in the body.
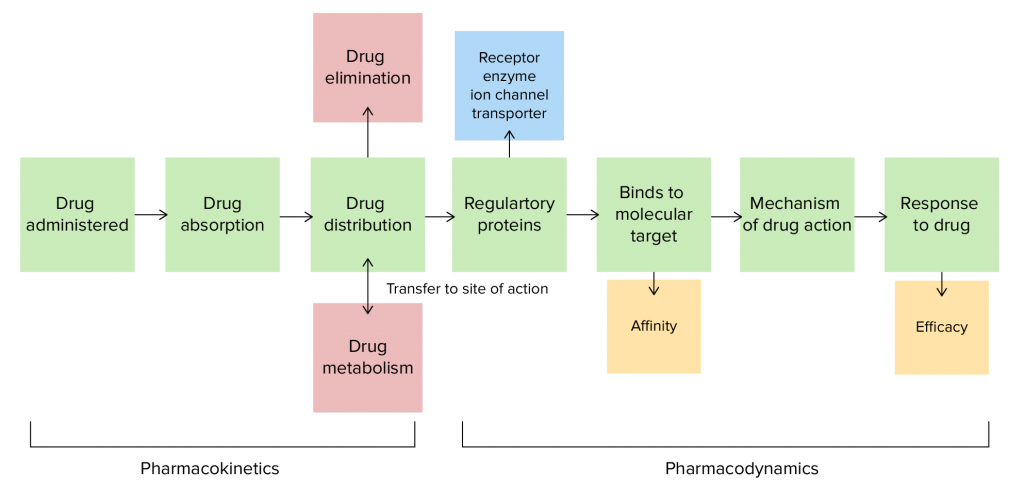
Many of these processes can be altered or function differently depending on our genetic makeup. This can be directly related to a better response to the same drug in two different people or, in other words, different efficacy in different people. It can also be associated with adverse effects, which may appear in some individuals and not in others.

But in addition, there are many other genes that are also associated with drug response: DPYD, NUTD15, HLA-B, CFTR, G6PD, TPMPT, etc....
Personalized medicine in a clinical setting
For this reason, many clinical consultations are starting to include this type of pharmacogenetic studies in those patients who are going to start treatment with a certain drug, for which it is known that there is a clinical recommendation. As we have commented in a previous blog, we know that the drugs used in the field of oncology and neurology are those for which there are a greater number of markers and clinical recommendations.
In order to standardize and normalize clinical recommendations, the international consortium for the clinical implementation of pharmacogenetics (CPIC) has developed guidelines for recommendations to facilitate their applicability in clinical routine. These guidelines are based on a literature review, creation of guidelines, publication in high impact journals, using standardized terminology. For this reason, nowadays they are the most recognized and widespread pharmacogenetic guidelines worldwide, being the one used in most laboratories and diagnostic centers in which pharmacogenetic studies are included.
Pharmacogenetics in ADNTRO
At ADNTRO we have just published your pharmacogenetic results based on CPIC recommendations for 27 different drugs. You can consult this list at the end of the article. However, this number may vary between users according to the SNPs that we have been able to analyze individually in each of the samples.
An example of one of the drugs we have included is Ibuprofen, a widely used drug, which is included in the Nonsteroidal Anti-inflammatory Drugs (NSAIDs). Ibuprofen acts by preventing the synthesis of prostaglandins and other prostanoids, by inhibiting the cyclooxygenase involved in inflammatory processes. Therefore, its main use is for the symptomatic treatment of pain and inflammation of traumatic or degenerative origin. For many people it is unknown that there is a gene associated with the response to ibuprofen, the CYP2C9 gene, which as we have shown in the previous table, is part of the P450 enzyme complex, and metabolizes some drugs including ibuprofen. For this reason, there are alleles associated with normal, intermediate or slow metabolism in this gene when it comes to ibuprofen.
Types of metabolizers - pharmacogenetics
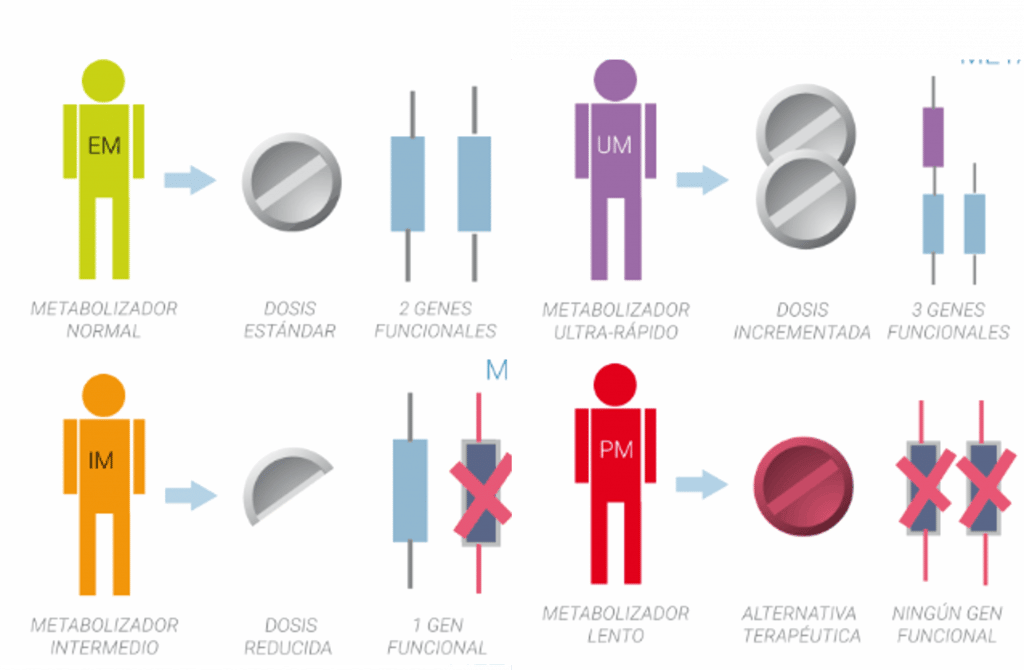
- CYP2C9 Normal metabolizer: Start treatment with the recommended dose.
CYP2C9 Intermediate metabolizer There are two recommendations depending on the enzymatic activity: Recommendation 1 Start treatment at the recommended dose. According to the prescribing information, use the lowest effective dose for duration compatible with each patient's treatment goals. Recommendation 2 Start treatment with the recommended dose. Increase the dose up to the maximum recommended dose clinical effect with caution. According to the prescribing information use the lowest effective dose for duration compatible with the patient's goals. Carefully monitor adverse events such as blood pressure and renal function during treatment.
- CYP2C9 Slow metabolizer: Initiate treatment with 25-50% of the lowest recommended starting dose. Adjust dose to clinical effect or to 25-50% of the maximum recommended dose with caution. Based on the prescribing information, use the lowest effective dose for the shortest possible time, consistent with each patient's treatment goals. The dose should not be increased until steady state has been reached (5 days for ibuprofen). Carefully monitor adverse effects such as blood pressure and renal function during treatment. Alternatively, consider alternative therapy not metabolized by CYP2C9 or not significantly affected by CYP2C9 genetic variants.
Log in and check your personalized pharmacogenetic recommendations within the health section.